Jingxu (Kent) Zheng, Qing Zhao et al. from Prof. Lynden Archer’s research group, report on a new concept that utilizes epitaxy to template the metal electrodeposition morphology in battery anodes, resulting in highly stable battery anodes that can operate over 10,000 cycles at ultrahigh rate.
In the latest issue of Science, appears a report from the Archer group describing a novel strategy to manipulate electrochemical and interfacial reactions in battery anodes that utilize metals such as Lithium, Sodium, Zinc, and Aluminum for low-cost & high-energy storage of electricity. In the paper, they show that the morphologies in which metals electrodeposit in battery anodes can be regulated by matching the lattice parameters and texture of the metal with those of the substrate. They utilize this discovery to build high energy density battery systems that remain stable for thousands of cycles of charge and discharge.
Replacing the graphite anode in present-day lithium ion batteries with a metal, including Li, Na, Al, Zn, etc., can theoretically increase the amount of energy stored per unit mass or volume of the anode by up to one order of magnitude. However, batteries based on metallic anodes are notorious for their propensity to fail prematurely during normal battery operation — upon charging, the metal cations in the electrolyte are reduced at the anode to form a solid metallic phase. This phase is ideally configured to be planar, but in most batteries it spontaneously transitions to create non-planar, irregular, morphologies that grow aggressivily to ultimately fill the inter-electrode space. This poses a serious barrier to practical implementation because it leads to active material loss via multiple mechanisms. It is also dangerous because the non-planar deposits can bridge the battery electrodes, short-circuiting the cell. The ohmic heat generated inside the cell by a short-circuit can lead to fire or explosion.
Researchers in the Archer group developed a general strategy using electrochemical epitaxy to overcome the problem of uncontrollable metal deposition morphology. In an epitaxial electrodeposition process, a thin-film electrodeposit forms a coherent lattice interface with the substrate. The single crystalline new phase (epilayer) exhibits a correlated orientation in relation to the substrate and low residual stresses. The strongest orientation correlations are achieved through directed nucleation and growth of the epilayer on a substrate that imposes minimal lattice strain. Textured interphases which add negligibly to the mass of a battery electrode and which form low-lattice-misfit interfaces are of specific interest as an epitaxial substrate.
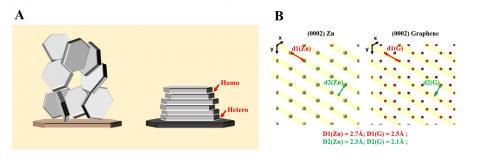
They selected Zn metal as a model battery anode to evaluate the concept of reversible electrochemical epitaxy. They found that Zn deposits formed on epitaxial substrates composed of graphene are well directed, showing a locked orientation relation with the substrate. This is in stark contrast to the deposition morphology on a traditional inert substrate, where Zn platelets are randomly oriented. Epitaxial Zn anodes achieve high reversibility (>99.7%) at moderate and high cycling rates.
Epitaxial electrodeposition as a strategy for creating highly reversible metal anodes is relevant for other electrode chemistries of contemporary interest. The authors proposed screening criteria for possible epitaxial substrates in other metallic anode systems, and demonstrated Au substrate for Al metal anode as an example.
To read the Cornell Chronicle Article: https://news.cornell.edu/stories/2019/11/zinc-anode-batteries-prove-the…;

